Hello, forum
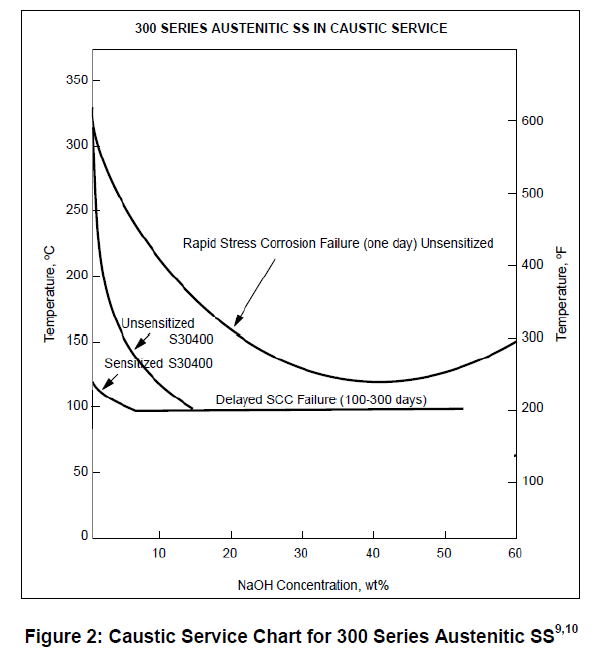
We are purchasing a plate heat exchanger (API 662-1) for water-water service. Materials of construction: plates 316L, shell CS. For hot side there is a short-term warm 15%w caustic case with risk of overheating up to 90°C which to be considered. Vendor does not want to provide PWHT for hot side and insists on 316L or A321 lining/cladding. Para. 4.5.3.6.b API 571-2011 states "300 Series SS offer little advantage in resistance to cracking over CS".
Can anyone share a worth source for CSCC of SS300?
We are purchasing a plate heat exchanger (API 662-1) for water-water service. Materials of construction: plates 316L, shell CS. For hot side there is a short-term warm 15%w caustic case with risk of overheating up to 90°C which to be considered. Vendor does not want to provide PWHT for hot side and insists on 316L or A321 lining/cladding. Para. 4.5.3.6.b API 571-2011 states "300 Series SS offer little advantage in resistance to cracking over CS".
Can anyone share a worth source for CSCC of SS300?