I'm trying to model a Resistance Spot Welding process using an implicit and transient thermal-electric problem on ABAQUS and came across this problem. Here's a simple test case in which i replicated the problem.
Consider the following axisymmetric analysis (no heat flows in or out of the system; any heat generated by the electric current stay within the system):
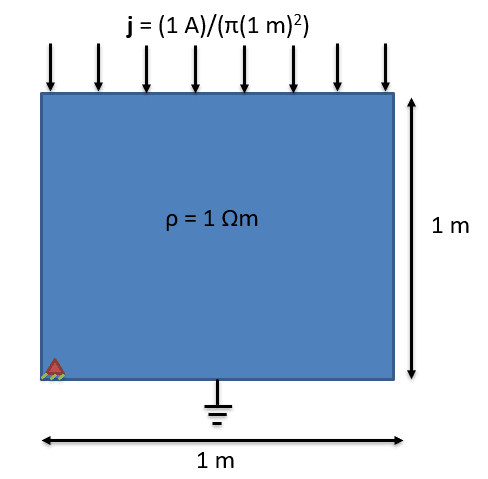
For a current of 1 A the current density is 0.318 A/m^2. For resistivity of 1 ohm-m, the resistance is 0.318 ohm. Consequently the voltage i get is also 0.318 V (IR = 1 A*0.318ohm).

The expected energy dissipated (let's call it 's') is (current_density)^2*resistivity*volume = 0.101*1*3.14 = 0.318 J
For a time-period of 1 s, I used the following fixed time increments to obtain the corresponding results:
Δt = 1 s : s = 0.105 J (one increment)
Δt = 0.5 s : s = 0.211 J (two increments)
Δt = 0.25 s : s = 0.264 J
Δt = 0.1 s : s = 0.296 J
Δt = 0.05 s : s = 0.307 J
Δt = 0.01 s : s = 0.316 J
Δt = 0.05 s - 1 s : s = 0.307 J (for automatic time control with initial time increment of 0.05 s)
Why does Δt = 1 s under predict the energy? Why is it that the first time increment always only output a third of total energy it should?
Any help is appreciated. Thanks.
Consider the following axisymmetric analysis (no heat flows in or out of the system; any heat generated by the electric current stay within the system):

For a current of 1 A the current density is 0.318 A/m^2. For resistivity of 1 ohm-m, the resistance is 0.318 ohm. Consequently the voltage i get is also 0.318 V (IR = 1 A*0.318ohm).

The expected energy dissipated (let's call it 's') is (current_density)^2*resistivity*volume = 0.101*1*3.14 = 0.318 J
For a time-period of 1 s, I used the following fixed time increments to obtain the corresponding results:
Δt = 1 s : s = 0.105 J (one increment)
Δt = 0.5 s : s = 0.211 J (two increments)
Δt = 0.25 s : s = 0.264 J
Δt = 0.1 s : s = 0.296 J
Δt = 0.05 s : s = 0.307 J
Δt = 0.01 s : s = 0.316 J
Δt = 0.05 s - 1 s : s = 0.307 J (for automatic time control with initial time increment of 0.05 s)
Why does Δt = 1 s under predict the energy? Why is it that the first time increment always only output a third of total energy it should?
Any help is appreciated. Thanks.
